Is the brain parasite Toxoplasma manipulating your behavior, or is your immune system to blame?
The immune system could be responsible of the change of behavior caused by brain parasite Toxoplasma.
We’re not the same when we get sick. Whether it is sneezing when we get a cold, or ferociously biting people when we get rabies, germs change our behavior.
That’s because germs need to transmit from one host to another. Consequently, host behavior is usually altered in ways that help the pathogen spread. Rabies, for example, causes infected animals to aggressively bite others because the virus transmits through saliva. But some microbes are more subtle.
Toxoplasma gondii parasites, which sexually reproduce only in cats but can infect any animal, hijack the brain and affect the host’s behavior. In a turn of events that would make Charles Darwin smile, rats and mice infected with Toxoplasma behave in ways that make them easy prey for cats – exactly where Toxoplasma wants to go.
The ability of Toxoplasma to disrupt such basic instincts in rodents is alarming when you consider that one-third of humans also carry this parasite’s cysts in their brain. Latent toxoplasmosis in humans has been associated with serious neurological disorders, including schizophrenia, intermittent explosive (rage) disorder and suicide, but has never been shown to be a direct cause.
Could the parasite be manipulating people as well? Is there a way we can get rid of this parasite and, if so, would behavior return to normal?
I am a microbiologist who has been studying Toxoplasma for over 20 years. Not only have I found the parasite’s effects on its host to be endlessly fascinating, I have been trying to identify its vulnerabilities so physicians can better treat this currently incurable lifelong infection.
In a collaboration with biochemist Ronald Wek and neuroscientist Stephen L. Boehm II, we have made the surprising discovery that the parasites may not be directly manipulating its rodent host. Rather, the host’s immune response to the chronic infection may be to blame.
Your brain on Toxoplasma
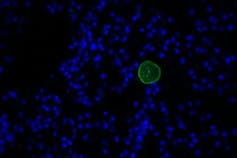 Toxoplasma cyst, green, in a section of brain tissue. The nuclei are stained blue.
Toxoplasma cyst, green, in a section of brain tissue. The nuclei are stained blue.Jennifer Martynowicz, CC BY-SA
Toxoplasma is a single-celled parasite that really gets around – it has managed to infiltrate the brains of billions of creatures around the world, from birds to beluga whales. Of all the species Toxoplasma can infect, though, only cats support its sexual stage. After sex in the cat gut, Toxoplasma is packaged into sturdy pods called oocysts that are released into the environment via feces, and can then be ingested or inhaled by other animals.
Infection with Toxoplasma does not usually produce symptoms in humans unless their immune systems are compromised, but the parasites remain in the body for life as latent tissue cysts. These tissue cysts are commonly found in the brain, heart and skeletal muscle. The formation of tissue cysts occurs in all infected animals, including many that end up on our dinner plate. Consumption of these tissue cysts in raw or undercooked meat also transmits the infection.
Another way these tissue cysts serve as a vehicle for parasite transmission is through the alteration of host behavior. Rats and mice with latent toxoplasmosis become hyperactive and lose their instinctual fear of cats, essentially making them a free lunch for felines.
Jennifer Martynowicz, an M.D.-Ph.D. student in my lab, was intrigued by the ability of latent toxoplasmosis to alter the behavior of mice. It has long been a mystery as to how exactly this little microbe, which seems inert when encased in its tissue cyst wall, manages to pull off such a feat. It is known that Toxoplasma releases an arsenal of parasite proteins into host cells that can alter gene activity, but how this translates into altering behavior remains unknown.
Previous work in our lab found that guanabenz, an FDA-approved drug used to treat hypertension, significantly reduces the number of brain cysts in an infected strain of mice we call BALB/c. Using this drug, Martynowicz was able to address a fundamental question: If we reduce the number of parasite cysts in the brain, can we restore normal behavior?
 Mice infected with Toxoplasma gondii are unafraid of cats.
Mice infected with Toxoplasma gondii are unafraid of cats.Fer Gregory/Shutterstock.com
Toxoplasma changes behavior – drug reverses it
Martynowicz administered guanabenz for three weeks to the mice that were hyperactive due to latent toxoplasmosis. When Martynowicz examined the brains of the treated mice and mice that received no guanabenz, she discovered that cyst counts were lowered about 75% in treated mice, reinforcing the results from prior studies.
In the first demonstration of its kind, Martynowicz then examined whether the reduction in cysts affected activity levels in the mice. To our delight, the hyperactivity usually seen in mice with latent toxoplasmosis had disappeared. The animals treated with guanabenz behaved like normal, uninfected mice.
So it looked like our lab’s hypothesis was correct: Brain cysts correlated with behavior changes.
To be certain that the hyperactivity was caused by the cysts, Martynowicz decided to examine the effect of guanabenz in a different mouse strain called C57BL/6, which are more susceptible to Toxoplasma. In this mouse strain, guanabenz did not lower cyst counts. But it reversed the hyperactive behavior. These unexpected findings showed that the hyperactivity in infected mice does not correlate with the number of parasite brain cysts after all.
To address this puzzling discrepancy, Martynowicz examined the level of inflammation in the brains of these mice. Other investigators have observed that latent parasite cysts in the brain recruit immune cells, producing a low level of sustained inflammation.
Is brain inflammation changing behavior?
Guanabenz is known to have anti-inflammatory effects. Decreasing brain inflammation is exactly what it appears to be doing in the brains of both infected mouse strains. These results suggest that the hyperactivity in infected mice is more likely driven by their immune response rather than a parasite-driven manipulation. If so, the key to controlling some behavioral changes in infected animals may be modulating their immune response.
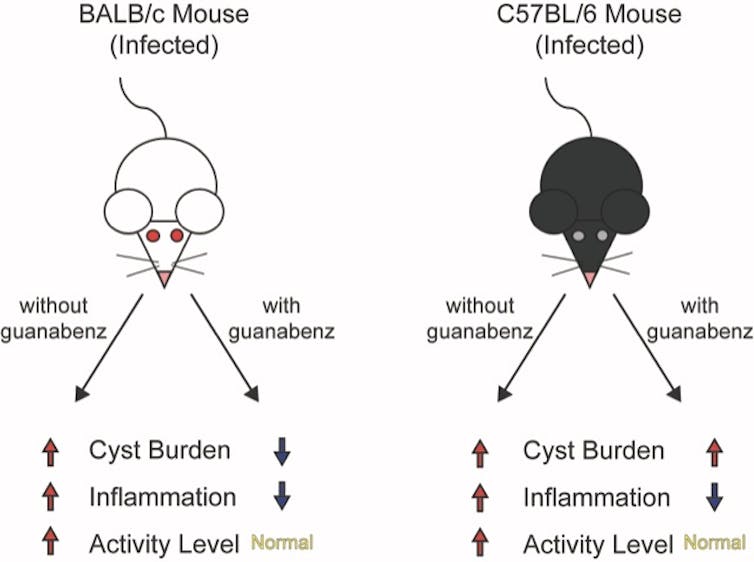 These two cartoons show the impact of guanabenz in two different strains of mice. The results suggest that brain inflammation may be to blame for behavioral changes.
These two cartoons show the impact of guanabenz in two different strains of mice. The results suggest that brain inflammation may be to blame for behavioral changes.Jennifer Martynowicz, CC BY-SA
We do not yet know how neuroinflammation may lead to hyperactivity. But it is interesting to note that some emerging studies have also found a link between inflammation and attention-deficit hyperactivity disorder (ADHD), which affects more than 6 million children in the U.S.
If our findings in mice, published in the journal mBio, extend to people, it could have important ramifications for how we currently treat brain infections. Our results suggest that brain infections may cause neurological consequences only in a subset of people, based on their immune response. Further studies are needed to determine if anti-inflammatory drugs like guanabenz may be effective at managing these conditions.
Bill Sullivan, Professor of Pharmacology & Toxicology, Indiana University
Este artículo fue publicado originalmente en The Conversation. Lea el original. Photo: fotovapl/Shutterstock.com, CC BY-SA

